6 Human Health Risk
This section discusses the evaluation of TPH at petroleum-contaminated sites to characterize human health risks. It also presents a discussion of the varying approaches to evaluating health risks for TPH, how the approaches need to be modified to address specific exposure routes, and how evaluation of metabolites and degradation products can be addressed. The discussion of how TPH risk assessment differs from the risk assessment of other chemicals and the variations in approaches to TPH risk assessment will serve as a basis for understanding and performing TPH risk assessment at individual petroleum release sites and can provide a basis for developing or revising regulatory approaches to human health risk assessment for TPH.
6.1 Introduction
Several methods are available for evaluating the human health risks associated with petroleum releases. One approach is estimating health risks for individual hydrocarbon compounds (e.g., BTEX, PAHs), and summing the risks and hazard indices estimated for each of the hydrocarbon chemicals evaluated. Such evaluations often address nonhydrocarbon components of the original mixture (e.g., additives such as MTBE) and help to estimate the cancer risk because TPH is not considered to be a carcinogen. These chemicals frequently make up a very small fraction of the bulk of the petroleum compounds for a release, which leaves uncertainty as to the health risk of the remaining mass. To address this issue, methods have been developed over the past twenty years to more precisely quantify TPH in terms of a small number of targeted, aromatic and aliphatic carbon ranges. Representative physiochemical constants and toxicity factors are then assigned to carbon ranges. This allows the data to be considered in standard risk assessments and related models in the same manner as is done for individual chemicals. Evaluating the human health risks associated with petroleum releases is the subject of ongoing research.
There are unique aspects of TPH that pose challenges when performing the risk assessment and interpreting the results. Three of the most important aspects of TPH that must be considered include the complex and variable nature of petroleum releases, the fact that the composition of any given released petroleum mixture can change substantially as a result of “weathering,” and the potentially substantial and selective partitioning of mixture components as they move from one environmental media into another. Over the last several years, regulatory concentration limits for TPH have evolved from being predominantly based on nuisance (e.g., odor, discoloration) to being increasingly based on human health protection Tomlinson and Ruby 2016. However, aesthetic considerations in addition to risk may need to be factored into environmental management decisions (see Case Studies).
Disturbance of heavily contaminated soil or groundwater during remediation and construction activities can also lead to the temporary but significant emissions of petroleum vapors and short-term risk to workers and occupants of neighboring properties. However, health effects are normally transient (temporary), and may quickly diminish once exposure is ceased. Methods to predict and manage shorter term risks are qualitatively discussed in Section 9 of this document.
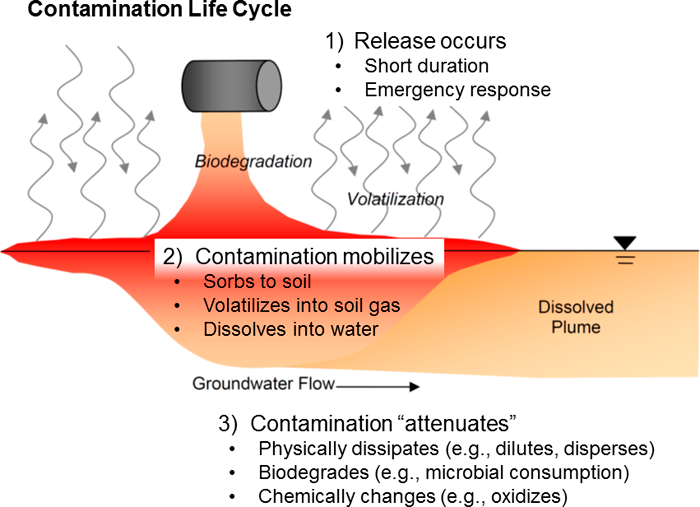
Figure 6‑1. Contamination life cycle.
Petroleum mixtures reported as TPH can consist of thousands of individual compounds, and mixtures reported as a specific petroleum product (e.g., gasoline) can have significantly different molecular compositions. Even greater variability can be expected for petroleum releases of crude or residual oil, and it is not unusual to have more than one refined product present at a release site. Not only is the originally released material at virtually all petroleum release sites a complex mixture of hundreds of hydrocarbons, but the composition of nominally similar materials (i.e., gasoline, crude oil, etc.) can differ in ways that would affect the environmental mobility and toxicity of the mixture (see Petroleum Chemistry and Refining).
Once released into the environment, chemical and biological processes will begin changing the composition of the originally released material. Although such “weathering” of released petroleum has long been recognized, the need to explicitly consider the chemical and biological degradation products in a human health risk assessment is a relatively recent development.
Most or virtually all of the mass of degradation products typically results from the oxidation of hydrocarbons. Accordingly, the degradation products tend to be dominated by polar compounds that have different chemical properties and behaviors in the environment and different toxic properties than their parent hydrocarbons. These degradation products are commonly referred to as metabolites, and largely for the sake of convenience, that term is used in the text below. Consideration of the metabolites is important because, while they are not necessarily present at all sites, they can make up the dominant mass of the residue from a petroleum release during specific phases of the lifecycle of the release (see Alterations of Petroleum Mixtures in the Environment through Weathering Processes).
A consideration in evaluating health risk is the fact that various constituents of TPH will tend to differentially partition from the main mass of a fresh or weathered release if they move from one environmental media to another. Surface water or groundwater affected by a petroleum release would be enriched in the more water-soluble constituents relative to the mixture that may have been released to soil, for example. Vapor intrusion is only likely to involve the more volatile constituents. Plant or fish uptake would also involve selective enrichment.
Some environmental media may not be amenable to TPH analyses. Concerns about health risks posed by accumulation of chemicals associated with a petroleum release in irrigated crops or fish or animal tissue could not be addressed by chemical analyses for TPH because the petroleum-related materials could not be distinguished from the plant or animal tissue. The risk assessor would need to decide on a set of appropriate indicator chemicals for such evaluations.
6.2 Exposure Assessment
Key factors to consider when assessing TPH exposures include the wide variation in physical and chemical properties of the mixture and potential exposure impacts as the mixture partitions between different environmental phases. Estimating exposure point concentrations in different media requires characterizing the TPH fractions (if a fraction-based approach is being used), individual chemicals, or both, that will be the basis of an evaluation. Any site-specific needs would also need to be considered, as would any applicable regulatory requirements or guidelines. Site-specific needs may also include consideration of acute effects and aesthetics (e.g., odor or visual impacts) (see Examination of Risk-Based Screening Values ITRC 2005, and Use of Risk Assessment in Management of Contaminated Sites ITRC 2008).
6.3 Direct Contact with TPH
Appropriate sampling and analysis methods for TPH in soil are presented in Section 5. The sampling methods need to consider the type of petroleum product, whether the product is new or weathered, and other environmental conditions (e.g., heat) that could affect the selection of complete exposure pathways or the quantification of exposure.
Direct contact with chemicals in soil typically involves ingestion, dermal contact, and inhalation of vapors or particulates USEPA 2017c. To evaluate these potential exposure pathways for a TPH risk assessment, the composition of the TPH mixture should be considered. For the ingestion and dermal contact pathways, bioavailability of TPH compounds may be considered. Although bioavailability methods for PAHs are not yet defined, the potential for refining the human health risk assessment (HHRA) may be considered. The use of bioavailability in risk assessment is discussed in the ITRC’s Bioavailability of Contaminants in Soil (BCS) document. Though the BCS document does not specifically discuss TPH, it presents methods for evaluating oral and dermal PAH bioavailability that can be applicable to TPH if individual PAHs are included in the HHRA. The BCS also discusses the considerations (e.g., PAH source and concentration, or skin models) for validating these methods and identifying other methods that can predict bioavailability with higher certainty.
For some TPH mixtures containing PAHs, dermal contact may contribute to the overall cancer risk USEPA 2018c. However, caution must be used when considering PAH dermal risk assessment because the EPA has not finished evaluating dermal toxicity data to develop appropriate dermal cancer toxicity values. In addition, methods have not been developed for the evaluation of direct contact with petroleum products based on total TPH (DRO/GRO) or TPH fractions. Currently, EPA has provided chemical-specific dermal absorption efficiency factors (referred to as AEd) for some TPH volatiles, and semivolatiles are assigned a default value of 10% USEPA 2004. EPA is continuously updating the dermal absorption values as information becomes available USEPA 2018c.
6.4 Soil to Ambient Air Exposure to TPH (Volatiles and Particulates)
Soil to ambient air inhalation exposure to TPH volatile components is commonly evaluated for individual TPH compounds (e.g., BTEXN and PAHs) using mathematical models, e.g., the Jury model Jury, Farmer, and Spencer. 1984, which predicts chemical transport and requires single-chemical data input (HLC, Kow, Koc), and dispersion models (AERMOD), which predict subsequent dispersion of vapors in ambient air. The Jury model does not accommodate mixtures USEPA 1996, 2002. However, where physiochemical property data and inhalation toxicity values are available for individual TPH fractions MADEP 2003, the Jury model can be used to estimate the flux for specified carbon ranges and the risk can be evaluated for specific carbon ranges. The risk to exposure to total (bulk) TPH can also be evaluated using weighted averages of the estimated risk for individual carbon fractions Brewer et al. 2013. It should be noted that exposure evaluation based on the Jury modeled flux cannot be used for evaluating soils when the TPH mixture is present as a free-phase product (saturated concentration) USEPA 2002.
Alternative methods for evaluating ambient air exposure to individual carbon ranges in soil that are used in many states include soil vapor and direct flux measurements. Direct flux measurements using flux chamber sampling are combined with dispersion modeling to estimate air concentrations that are then compared to ambient air screening levels of individual compounds or petroleum hydrocarbon mixtures USEPA 2002; ITRC 2008.
For particulate emissions of TPH in soil, the particulate emission factor (PEF) can be used, which is applicable to all types of TPH compounds, because it is not chemical-specific. At sites with increased soil-based activities (e.g., construction sites), the particulate emissions due to vehicular traffic on unpaved roads should be considered in the PEF derivation USEPA 2002; ITRC 2008.
6.5 Leaching from Soil to Groundwater
As discussed in Sections 5.3 and 5.4, physiochemical constants assigned to individual carbon ranges can be used in soil leaching models to derive leaching-based screening levels or predict groundwater concentrations resulting from soil migration to groundwater in the same manner as carried out for individual compounds. To estimate the potential for TPH mixtures to leach to groundwater, it is important to incorporate appropriate chemical-specific values (HLC, Koc) specific to each TPH fraction USEPA 1996. The accuracy of commonly used leaching models is dependent on site-specific parameters and adequate knowledge of the physio-chemical properties of the contaminant(s). This can be particularly challenging for complex mixtures such as TPH. Further research regarding this subject is required.
In general, the potential for leaching will be greatest for the <C9 aromatics and other compounds with high aqueous solubility (e.g., TPH polar metabolites). The leaching potential of polar metabolites has yet to be evaluated. Identifying and incorporating mole fractions of the petroleum hydrocarbon mixture into the leaching evaluation is essential. Failure to include TPH fractions in this pathway’s evaluation can result in inaccurate estimates of release from soil and transport into the water phase, which may be amplified when (1) the TPH mixture is present as LNAPL; or (2) the petroleum hydrocarbon mixture contains heavier oils (higher molecular weight compounds) with negligible aqueous solubility.
Analytical tools used to empirically determine TPH leaching potential include the Synthetic Precipitation Leaching Procedure (SPLP) analysis (EPA SW-846 Method 1312) and the SPLP Batch Test Leaching Model, which both use a slightly acidic extraction fluid to estimate leachate concentrations in situ.
If LNAPL is present in soil, leachate analyses may inaccurately predict the leaching potential of the TPH mixture due to direct partitioning of nonsorbed droplets into the leachate solution. One option to help minimize this uncertainty is the use of soil column leaching tests. See Case Studies 1, 2, 3, and 5 for more information. Note that polar metabolites can form in the vadose zone as hydrocarbons migrate.
6.6 Exposure to Groundwater/Surface Water
Petroleum spills and leaks to land surface or surface water bodies can result in elevated concentrations of TPH and TPH-related metabolites in groundwater or surface water via the migration of free product or leaching. Short-term (subchronic) and long-term (chronic) exposures to TPH can occur via contact with groundwater used as a potable water supply (e.g., private drinking water wells) and other water uses (e.g., swimming pools). TPH presence in groundwater can be detected by affected receptor populations from the distinctive taste, discoloration, and odor characteristic of some of the individual TPH components (e.g., benzene).
Due in part to the complexity of TPH components themselves, determining their potential exposures becomes a challenge. The exposure point concentration is heavily dependent upon the specific analytical method details and requirements, which vary from state to state. Over the years, the selection of surrogate or indicator compounds (or a combination of compounds) to represent TPH has become a common remedy to this problem.
Exposure to surface water may be lower than that of groundwater due to generally less frequent contact and shorter term average exposures or shorter average exposure durations. In instances where TPH-contaminated groundwater may be discharging to a surface water body, some states allow use of surface water modeling and site-specific dilution factors for high flow, lentic systems to predict TPH concentrations in surface water. Other state agencies, such as MADEP, have issued policy in the form of a support document on the VPH/EPH approach to modeling MADEP 2002b.
A number of challenges exist when investigating and addressing the risk from exposure to fuel-related chemicals in surface water bodies (e.g., rivers, creeks, sensitive wetlands, drainage canals, ditches, roadway runoff, etc.) that may be flowing or discharging to other locations. One issue is the presence of competing sources; the collection of background data can be critical in eliminating these other sources. In addition, some surface waters (e.g., small wetlands) may have normally high background concentrations of nonpetroleum-related compounds detectable in the TPH analysis that must be distinguished from potential sources in question. High background levels may come from either naturally occurring sources (e.g., natural seeps) or anthropogenic sources. Environmental forensics could help in making this distinction between the footprint of TPH in background material and TPH from a particular source.
6.7 Exposure to Air
6.7.1 Outdoor (Ambient Air)
Inhalation toxicity factors assigned to individual carbon ranges can be used to develop risk-based screening levels for ambient air and/or directly quantify human health risk in the manner as done for individual compounds. Exposure assessment of TPH and TPH-related compounds in ambient air resulting from emissions from contaminated soils can be complicated and, in many cases, irresolvable in the presence of multiple (non-site-related) sources of TPH in ambient air, including fueling stations, auto exhaust, cleaning fluids, and other uncertain and nonpoint sources. Because of these potentially confounding sources, it may be important to include sufficient background (or reference) locations in any outdoor air TPH sampling program, if ambient air exposure is to be measured directly. The alternative to direct measurement, modeling of ambient air concentrations from soil, is discussed in Section 6.4
6.8 Food Chain
Due to the limited studies, bioassays, and heightened uncertainty in the bioaccumulation and biomagnification of TPH in aquatic and terrestrial ecosystems, these evaluations are typically not recommended for use in risk management decision making. Instead, individual chemicals of concern become the focus. In general, the lower molecular weight aliphatics and aromatics do not bioaccumulate, but there is some evidence that aquatic and terrestrial organisms do bioaccumulate some TPH constituents, particularly PAHs Farrington et al. 1982. However, depuration does occur if the source of the contamination is removed Cox et al. 1975; Williams et al. 1989.
Some of the difficulties associated with trying to evaluate the food chain uptake pathway include the following:
- terrestrial plants and aquatic algae can metabolize hydrocarbons, making it difficult to decipher whether the hydrocarbons in the soil will actually be in the plants from the soil Eisler 2000
- plants and animal tissues are made up of organic chemicals, which will often themselves report as TPH if analyzed Hoffman et al. 2003
- models can attempt to predict only what could actually occur (e.g., use of food chain models that estimate uptake may not truly reflect what is actually happening). Similar arguments can be applied to the animal meat uptake pathway (see Ecological Risk Assessment).
6.8.1 Plant Consumption
Humans may be indirectly exposed to TPH via plant ingestion in instances where food crops are irrigated with water containing TPH, for example. However, because TPH analytical methods would extract plant material as well as any petroleum components, they cannot be used to analyze for petroleum residues in plants. Risk assessment of TPH in plant material must rely on chemicals selected to match the surrogate chemicals of potential concern in the soil or irrigation water to which the food crops may have otherwise been exposed. Further information on plant uptake of TPH and TPH toxicity in plants is provided in Ecological Risk Assessment.
6.8.2 Meat and Milk Consumption
Human exposure to TPH may also occur through the ingestion of livestock (e.g., contaminated meat) or consumption of milk from animals that may graze on TPH impacted soil or ingest TPH-contaminated water as drinking water. As with plant consumption, TPH analytical methods are not applicable to meat or milk. Indicator chemicals relevant to the spilled petroleum that would be expected to partition into and possibly accumulate in the edible portions of the target species need to be selected.
6.8.3 Fish and Shellfish Consumption
Consumption of fish and other aquatic organisms following a fuel release or crude oil spill into surface water is an exposure scenario that often needs to be addressed. Fisheries may be closed as a preventive measure following a spill, and a human health risk assessment may be required before the fishery can be reopened. As with plant and other animal tissues, predicting or measuring the uptake of hydrocarbons into fish tissue from a specific petroleum release is challenging. Chemical analysis of fish and shellfish using fractionation or a bulk TPH method is not practical because fish oil and other tissue components will be detected as TPH. Rather, indicator chemicals relevant to the spilled material that would be expected to partition into fish and shellfish need to be selected Yender et al. 2002; CAOEHHA 2015. In addition to a human health risk assessment, it may be necessary or desirable to perform a sensory evaluation (i.e., testing for off-flavors and off-odors) of the fish and shellfish in support of a decision to reopen a fishery Reilly and York 2001. Although analytical instrumentation is becoming more sophisticated, these instruments are chemical detectors only and are incapable of making any sensory judgment on odor or flavor NOAA 2001.
6.9 Toxicity Assessment
Equally important as exposure assessment in risk evaluation is the understanding of toxicity. The section below presents some focused background points for the discussion of the toxicity assessment of TPH along with a discussion of the basic approaches that have been taken to the toxicity assessment of TPH. The challenges and proposed approaches to evaluating the toxicity of petroleum metabolites are also discussed.
6.9.1 Background and Overview of TPH Toxicity Assessment Methods
There are a few basic approaches to toxicity assessment for TPH. Each approach addresses the challenges posed by the assessment of TPH toxicity in a different way. The best approach to apply in any given situation, however, is often a hybrid approach, particularly if multiple exposure routes need to be addressed.
The discussion of toxicity assessment for TPH presented below focuses on methods for evaluating chronic exposures, but acute exposure can also be a concern for TPH releases. Information relevant to addressing acute exposures to the public and in the workplace has been provided most directly by the ATSDR, the Occupational Safety and Health Administration (OSHA), and the National Institute for Occupational Safety and Health (NIOSH).
6.9.2 Approaches to TPH Toxicity Assessment
Recognizing the challenges posed by the need to characterize the toxicity of TPH, a variety of approaches have been developed and are currently in use. The oldest approaches rely primarily on an evaluation of indicator chemicals, which may or may not have been accompanied by an evaluation for various whole product fractions (e.g., gasoline range total petroleum hydrocarbons, diesel range petroleum hydrocarbons, or residual range total petroleum hydrocarbons). More recently, a variation of the indicator chemical approach has evolved, in which the bulk TPH mixture is divided into its aliphatic and aromatic components and then into discrete carbon ranges smaller than that of a typical petroleum product. The potential toxicity of these fractions is then used to evaluate risk, generally in conjunction with an evaluation of selected individual hydrocarbon chemicals. These general approaches are illustrated in Figure 6-2, which is from the TPHCWG 1999.
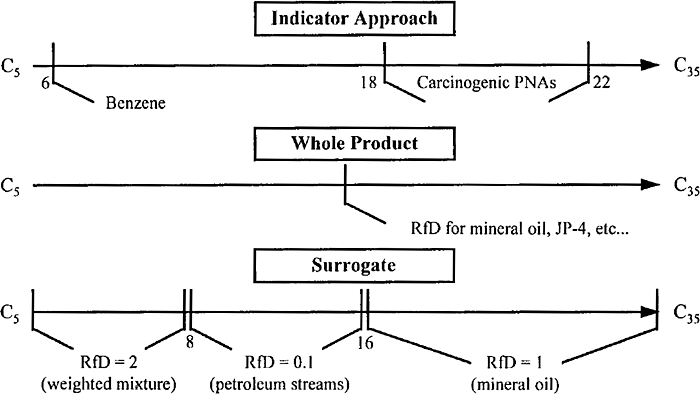
Figure 6‑2. TPH toxicity assessment methods TPHCWG 1999.
The three basic approaches to toxicity assessment of TPH are described below, including the indicator chemical approach, the fractionation approach, and whole product approach. Although each of these basic approaches are distinct, they are most often used in conjunction with each other (see TPH Fundamentals).
6.9.2.1 Indicator Chemical Method
One approach to evaluating health risks for petroleum hydrocarbon mixtures is to rely on indicator chemicals USEPA 1986, 1989, 2000c. Local guidance can vary for the indicator chemicals to be used for either cancer or noncancer health risks. The results of the States Survey show the indicator chemicals recommended by several different states.
The assumption underlying the use of the indicator chemical approach is that the indicator chemicals represent most of the mixture’s toxicity, and therefore, the health risk of the entire mixture can be estimated to a reasonable degree of certainty by relying on the health risks estimated for the indicator chemicals USEPA 1989. Contrary to this assumption is the fact that the indicator chemicals often make up a small fraction of the mass of the TPH range to which they are applied. On the other hand, the indicator chemicals are thought to be more toxic than the rest of the chemicals in the mixture. Concern over the validity of the indicator chemical approach is most often raised for sites with weathered releases of products where the indicator chemicals used for the released product(s) may not be present at detectable levels, but at which residual hydrocarbons are noticeably or measurably present. In such cases, reliance on indicator chemicals alone can cause health risks to be underestimated or can leave questions about the adequacy of an evaluation relying solely on indicator chemicals.
6.9.2.2 Fractionation Methods
Under the fractionation approach TPH is divided into aromatic and aliphatic compounds, and the aromatic and aliphatic compounds are further divided into several defined carbon ranges. The various fractions were originally divided into groups that would have similar fate and transport properties MADEP 1994, 2002a; TPHCWG 1997a. Massachusetts MADEP 1994, British Columbia BCMoE 1995, and the TPHCWG 1997a were the earliest groups to develop and recommend use of the fractionation approach. The ATSDR 1999, USEPA 2009d, and several states have also adopted versions of the fractionation approach. Examples of fractions and the toxicity factors assigned to them are shown below in Tables 6-1 and 6-2. Variations in the fractionation approach include different divisions of the fractions to be addressed and different approaches to the assignment of a toxicity value to each fraction (e.g., assign the toxicity of a constituent typical of the toxicity of the fraction or assign the toxicity of the most toxic constituent).
Table 6‑1. Summary of noncancer oral toxicity values (mg/kg-day)
TPHCWG 1997c; MADEP 2003; USEPA 2009d; TCEQ 2010
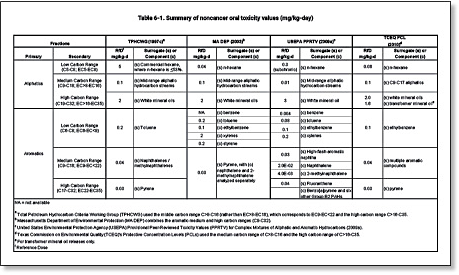
View Table 6-1 in Adobe PDF format.
Table 6‑2. Summary of noncancer inhalation toxicity values (mg/m3)
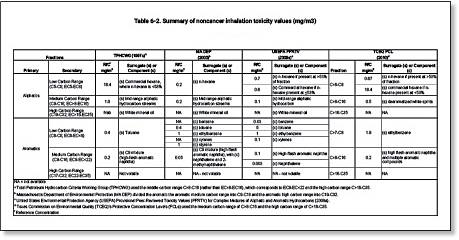
View Table 6-2 in Adobe PDF format.
6.9.2.3 Whole Product Method
Under the whole product approach, a single toxicity factor is assigned to a whole petroleum product, such as gasoline, diesel fuel, jet fuel, bunker fuel, crude oil, etc. A variety of approaches may be used to assign the toxicity factor for the product. For example, toxicity test results from testing a whole product might be used. The primary drawback to this approach as discussed above is the fact that the composition and toxicity of the product will change as it weathers and could only be applied to fresh releases. Another important consideration is the higher degree of variability in the unrefined petroleum materials (e.g., bunker fuel and crude oil), which would pose a great deal of uncertainty in extrapolating toxicity test results from one tested product to others. In addition, test results are not available for all of the whole petroleum products.
Because whole product TPH analytical results are frequently a significant portion of, if not the only TPH data available for a site, one can be faced with having to evaluate health risk for such reported mixtures. Although a variety of approaches can be used to assign toxicity factors to whole product carbon ranges, appropriate regulatory guidance on the topic should be consulted for recommended approaches. The most technically defensible approach is arguably to make assumptions about the proportions of TPH fractions comprising the released petroleum mixture and use recommended toxicity factors and chemical constants for the assumed fractions. While this approach may be the most technically defensible, it nonetheless entails more uncertainty than is associated with health risk estimates based on measured TPH fraction data. Use of this approach allows at least a screening level evaluation of whole product data. Two agencies have published guidelines for this method of evaluating health risks for whole product TPH (e.g., GRO, DRO, RRO) CASWB-SFBR 2016a; HIDOH 2017.
The whole product method of evaluating the toxicity of TPH entails more uncertainty than is associated with health risk estimates based on measured TPH fraction data. It can have value as part of a screening level evaluation, but should be used with great caution, if at all, for human health risk assessments. Local guidance should be consulted before using a whole product method for evaluating the toxicity of TPH.
6.10 Evaluating Toxicity of Polar Metabolites
This section discusses the challenges to evaluating the toxicity of the polar metabolites and the approaches that have been proposed for addressing the challenges.
6.10.1 Challenges in Evaluating the Toxicity of Polar Metabolites
It is well established that hydrocarbons (and other chemicals) undergo “weathering” (see Alterations of Petroleum Mixtures in the Environment through Weathering Processes) in the environment. Understanding the toxicity of the resulting hydrocarbon degradation of products is important because the majority, if not all, of the hydrocarbons at a petroleum release site can be present in soil or groundwater in the form of polar metabolites. Although much, if not all, of the mass of the metabolites can degrade to carbon dioxide and water eventually, the polar residues can be the source of ongoing potential or actual exposure for varying periods of time after a release. Accordingly, explicit evaluation of the metabolites may be warranted when they are present.
6.10.2 Approaches Proposed for Evaluating the Toxicity of Polar Metabolites
Two specific approaches for evaluating the toxicity of polar metabolites have been proposed. One assumes that the bulk polar metabolites have the same toxicity as the bulk hydrocarbons in the TPH fraction in which they are detected CASWB-SFBR 2016b. A second approach assigns toxicity factors for individual polar oxygenate degradation products to chemical structural families of degradation products Zemo, O’Reilly, et al. 2013; Zemo et al. 2017. Consideration of these approaches is discussed in the TPH case studies published by HIDOH 2018.
Both of these approaches are primarily intended to address refined middle distillate products in water. The focus on the middle distillate range is primarily because gasoline range organics are generally addressed by the evaluation of relatively water-soluble indicator chemicals (i.e., BTEX) and because polar metabolites of gasoline range hydrocarbons tend to have retention times in the middle distillate range. In addition, hydrocarbons larger than those in the middle distillate range from refined products have such low water solubility that they are not found dissolved in water at significant levels. One concern associated with both of these approaches is that the methods for measuring TPH-d will not always detect all of the polar compounds present in a sample and that the level of polar compound can be underestimated Bekins et al. 2016; Mackay et al. 2018.
6.11 Toxicity Considerations for Different Exposure Routes
Any uptake of hydrocarbons into plant or animal material will also involve a substantial fractionation of the hydrocarbon mixture with only a subset of the hydrocarbons being absorbed into the plant or animal and distributed to the edible portions. As noted above, TPH extraction methods will extract chemicals other than hydrocarbon from plant and animal material. Accordingly, TPH analyses cannot be used for the evaluation of hydrocarbon exposure from consumption of plant and animal material. An indicator chemical approach is required instead.
Consideration of the site-specific hydrocarbon mixture to which plants and animals are exposed and the mechanism by which they may take up the hydrocarbons will be important in the selection of appropriate indicator chemicals. For plant and animal exposures involving migration or solution into water as a first step, one would need to consider the fact that hydrocarbons soluble in water would be expected to be limited primarily to aromatic compounds with carbon sizes of approximately C14 and lower, and that larger aromatic structures and almost all but the smallest aliphatic structures have limited solubilities in water.
As with hydrocarbons, different portions of the degradation product mixture can be expected to partition to different environmental media, based on their chemical and physical properties. When evaluating exposure via inhalation (e.g., from vapor intrusion), the chemical constituents expected are generally limited to the most volatile constituents of a released petroleum mixture. The mixture may include degradation products (e.g., methane), but is not likely to include polar metabolites. Selecting a hydrocarbon representative of the volatile hydrocarbons expected in indoor air and soil gas could address the toxicity of the parent hydrocarbons, as well as any degradation products for the inhalation exposure pathway.
When considering potential exposure from water, gasoline range compounds are thought to be essentially limited to soluble hydrocarbons and not to include polar metabolites, which are expected to have retention times that would be detected in the middle distillate range of a TPH analysis. TPH measured in water originating from releases of middle distillate products (e.g., diesel fuel, Stoddard solvent, heating oil, jet fuel, etc.) into water can be present in water largely, if not entirely, in the form of polar degradation products. This is a function of the polar chemical structures, which are inherently more soluble in water than hydrocarbons. Hydrocarbons soluble in water from these products would be expected to be limited primarily to aromatic compounds with carbon sizes of approximately C14 and lower. Larger aromatic structures and almost all but the smallest aliphatic structures have limited solubilities in water.
Although it has been shown that polar degradation products can form in soil Mao et al. 2009a, general assumptions about the mix of metabolites and the fraction of material detected as TPH that is parent hydrocarbon versus metabolites are particularly elusive. Several factors that can affect the composition of polar degradation products following a petroleum release were identified (e.g., specific hydrocarbon product(s) released, chemical and microbial conditions in soil, time since release(s), etc.). It was also noted that a significant fraction of the material detected as TPH in soil samples analyzed by routine TPH analytical methods can be natural organic components of soil, particularly for organic-rich soil types, such as peat. The presence of such natural, organic soil components complicates any attempt to separately characterize the petroleum metabolites and parent petroleum compounds from the mixture of substances making up the TPH measured in soil samples.
It is plausible that TPH degradation products could be taken up in the food pathway as a result of irrigating food crops with water containing petroleum or as a result of uptake into fish from surface water releases, for example. It is not clear, however, which mixtures of degradation products or which individual degradation products would be taken up into a food exposure pathway, and thus, it is not clear what toxicity factors would be representative of any polar degradation products taken up into plants or animals in the food chain. Because many of the naturally occurring organic compounds making up plant and animal materials would report as TPH when measured using routine (bulk) TPH analyses, these analyses would not provide a valid measurement of the level of petroleum hydrocarbons or polar degradation products in plant or animal food products. Food pathways are typically evaluated using indicator chemical approaches. At some time in the future, it may be possible to select site-specific indicator chemicals for polar degradation products, but this is not currently possible using the best available scientific methods.
6.12 Risk Characterization
Risk characterization is the final step of the human health risk assessment process, and it has been described as the bridge between risk assessment and risk management because it provides a basis for risk estimation and an understanding of the results and uncertainties that are inherent in each step of the evaluation. ITRC 2015 described key issues associated with risk characterization and organized these into topic areas of risk results, presentation of risk results; and uncertainty and bias. More information on risk characterization is also available on USEPA’s Human Health Risk Assessment web page (USEPA 2017c Step 4, Risk Characterization).
Major assumptions, scientific judgments, and to the extent possible, estimates of the uncertainties embodied in the risk assessment are presented in the risk characterization. For risk assessments involving TPH, the understanding and description of uncertainty and bias can be even more challenging because TPH is often viewed as a “complex mixture” that is unique from other complex mixtures (e.g., Aroclors). The approach to characterizing the risks from TPH may not be entirely unique, although many assumptions associated with the measurement, fate and transport, toxicity, and risk estimation for TPH are unique and will vary depending on the specific approach being taken to TPH in any given risk assessment. Identification and discussion of how the TPH-specific uncertainties can influence results and conclusions is an important component of any TPH risk assessments.