7 Ecological Risk Assessment
This section provides practical guidance on addressing the unique challenges associated with conducting an ecological risk assessment (ERA) for TPH. Although an ERA may be warranted at TPH release sites with potentially complete exposure pathways for ecological receptors (see Conceptual Site Models), the approaches that have been used for TPH ERA vary by state and country with no single widely accepted TPH ERA guidance.
This section also presents tiered approaches for TPH ERAs, ranging from simple and qualitative to complex, site-specific, quantitative ecological risk evaluations. It describes types of TPH analytical data that may be appropriate for ERA, ranging from whole products to fractions to individual constituents, and provides a framework for selecting analytical data that are appropriate for the exposure media and receptors. This section also describes both model-based and toxicity testing–based methods for developing threshold toxicity values for TPH. Options and best practices for risk characterization are presented, as well as a practical overview of issues to consider and pitfalls to avoid in TPH ERAs. The proposed approach is generally consistent with USEPA guidance for ecological risk assessment USEPA 1997a.
7.1 ERA Process for TPH
TPH releases and exposures can range from single-event, petroleum product releases (e.g., crude oil spill into a marsh) to long-term discharges (e.g., diesel-contaminated groundwater seeping into a creek). ERAs typically evaluate a range of habitats, trophic levels, and, most importantly, representative species. Theoretically, each species could have a different response to TPH exposure. This, coupled with the fact that TPH is a complex mixture of numerous compounds that can change through time via physical weathering and/or biological transformation, makes TPH-focused ERAs particularly challenging.
The ecological effects of short-term and long-term releases are typically very different. During an accidental, single-event petroleum product release, the smothering of invertebrates or a reduction in the insulating properties of avian feathers is likely to be the primary risk mechanism in terms of immediate effects. Long-term discharges of TPH components to surface water are likely to have chronic effects on the receiving aquatic community that may be subtle and harder to quantify (relative to a petroleum product release such as a crude oil spill). This section focuses on the toxicological effects of short-term or long-term discharges rather than the physical effects associated with oil spills.
Additional information is available from National Oceanic and Atmospheric Administration (NOAA) guidance (https://oceanservice.noaa.gov/hazards/spills/) and other relevant materials, such as the International Oil Spill Conference Proceedings website (http://ioscproceedings.org), to address short-term spills, acute exposures, and their immediate aftermath.
Finally, elevated ecological risks in the narrow context of TPH toxicity do not always warrant remediation efforts that may themselves cause severe ecological disturbance or habitat destruction. Methods, such as Net Environmental Benefit Analysis, may be used to decide on the appropriate remedial strategies that will reduce risk and minimize collateral damage API 2013; USEPA 2006a
7.2 Current National and North American TPH ERA Practice
At this time, there is no national guidance for TPH ERA in the United States. However, TPH ERA screening guidance has been published by a few states and in Canada. The TPH States Survey indicates that 17 states follow publicly available guidance for conducting TPH ERAs. Of these, only New Jersey, Hawaii, Washington State, and California State Water Boards—San Francisco Bay Region (CASWB-SFBR) have developed ecologically based TPH screening levels (Table 7-1). Typically, these states have designated specific analytical methods to obtain TPH-based data (see TPH Analytical Methods and States Survey). Table 7-1 also provides TPH ERA screening levels for USEPA Region 4 and Canada.
7.3 Exposure Assessment
This section describes the variety of factors that should be considered in exposure assessment for TPH ERA. It includes selection of appropriate TPH analytical data that can be used in the screening and site-specific tiers, the distribution of different TPH fractions among various environmental media, and their bioaccumulation potential. As part of both the screening and site-specific tiers, this section also describes approaches for identifying ecological receptors, their routes of exposure to TPH, and exposure dose estimation.
It is important to note that potential ecological exposure does not occur for all releases. For example, releases to areas that have no viable habitat (e.g., paved or gravel areas, manufacturing areas with minimal ecological receptors) and no potential for off-site transport to adjacent habitat areas likely do not warrant consideration for potential ecological risk (see Table 5-3).
7.4 Toxicity Assessment
Toxicity assessment for ERAs can be challenging and highly variable because the assessment must consider toxicity to a wide range of receptors and groups and extrapolate toxicity data to protect populations or communities rather than individuals (except for specific cases). Additionally, for TPH, there is no single source of peer-reviewed or agency-approved toxicity values for ecological receptors, which, in part, stems from the complexity of TPH toxicity as discussed below.
Reference toxicity values for ecological receptor communities are based on both No Effects and Lowest Effects levels USEPA 1997a. For plants, invertebrates and fish are typically reported as lethal concentrations (LC) and sub-lethal effective concentrations (EC) that affect some percentage of the exposed population, LC and EC refer to the exposure concentration of a chemical at which a health response is noted in the specified percentage of the population, e.g., mortality in 50% of test fish (LC50).When more protective values are desired, the LC or EC values may be reported to a lower percentage of the exposed population such as 10% or 25% (e.g, LC10, EC25). When reference toxicity values are reported for higher level receptors such as birds and mammals, the exposure dose is referred to a No Observed Adverse Effect Level (NOAEL) or Lowest Observed Adverse Effect Level (LOAEL).
The USEPA and most states do not specify a single recommended toxicity value for TPH and the associated chemicals for ecological risk. However, compilations of studies and toxicity values are available in various databases such as EcoTox and the Risk Assessment Information System, and the agencies may select certain values as the basis for the development of water quality criteria or ecological soil screening levels.
When available toxicity data are insufficient or inappropriate for use for risk assessment of TPH at a given site, product-specific or site-specific toxicity testing can provide a means to assess a site’s unique TPH mixture or species sensitivity. However, toxicity testing of petroleum compounds has many limitations and technical challenges Singer et al. 2000; Redman and Parkerton 2015 that can confound results, and the presence of non-TPH chemical constituents or other stressors (e.g., nutrients) in site media can complicate site-specific testing. As an alternative, mechanistic models provide a means to develop toxicity thresholds or evaluate risk using site-specific conditions without some of the challenges of toxicity testing (see Overview of Toxicity Prediction by Mechanistic Models).
Figure 7-1 illustrates the two most common categories of methods for developing ecological toxicity threshold values for TPH and their associated uses: thresholds derived from mechanistic toxicity prediction models (which have been validated by laboratory test results), and thresholds derived from toxicity tests reported in the literature or from site-specific testing. Both model-based toxicity values and toxicity test-based toxicity values can be valuable in TPH ERA.
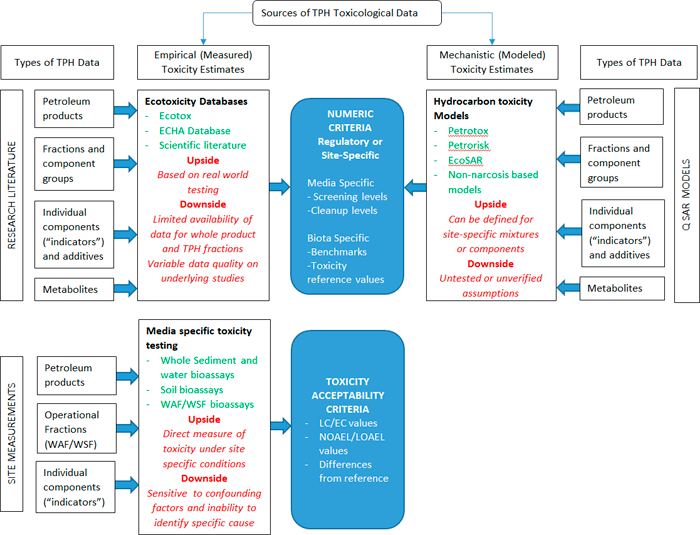
Figure 7‑1. Sources, types, and uses of TPH toxicological data for ERA.
(Source: B. Bjorkman, GEI Consultants, 2018.)
7.4.1 Overview of TPH Toxicity to Various Taxa
TPH toxicity to ecological receptors depends on its hydrocarbon composition as well as the exposure pathway and duration of exposure Albers 2003. Additionally, TPH in the form of product (e.g., crude oil, heavy fuel oil) can cause physical and chemical toxicity. Toxicity associated with exposure to high concentrations of TPH typical of an accidental release is both physically and chemically mediated and is very different from the subtle and lower magnitude of effects associated with exposure to chronic low-level releases, which is primarily the result of chemical toxicity. Because this guidance is focused on evaluating the risk of the TPH fraction of petroleum and not the petroleum product itself, the focus of this section is on the chemical toxicity of TPH and the more extensively studied mechanisms of chemical toxicity upon which mechanistic toxicity models are based.
Examples of toxicity end points of TPH, TPH fractions, and operational fractions for ecological receptors are summarized in Tables 7-5 and 7-6. This table highlights some of the reported end points, many of which are the basis for screening-level values and toxicity reference values, but is not a comprehensive list of all relevant toxicological end points or all end points reported in the literature. For the purposes of this table, WAF, WSF, and chemically enhanced water-accommodated fraction (CEWAF) are defined as described in Adams et al. 2017. The water-accommodated fraction (WAF) is generated by low-energy mixing of oil floating on water, with little disturbance of the surface slick. Test solutions contain dissolved hydrocarbons and potentially a small fraction of particulate oil. The water-soluble fraction is WAF that has been filtered or treated to remove all oil droplets and contains only truly soluble fractions of each test oil. CEWAF is generated by mechanical and chemical dispersion of floating oil. Test solutions contain dissolved hydrocarbons and a significant amount of particulate oil.
7.4.2 Mechanisms of Chemical Toxicity of TPH
The major mechanisms by which TPH compounds are understood to exert toxicity to ecological receptors include narcosis and phototoxicity. The chemical toxicity is considered to be additive in nature and described in more detail below.
7.4.3 Overview of Toxicity Prediction by Mechanistic Models
Predictive tools to estimate the ecological toxicity of TPH have been developed and used by regulatory agencies as well as professional and academic organizations. In general, predictive approaches use a combination of the physical and chemical properties of the chemicals and an assumed narcosis mode of action to predict the toxicity of a particular constituent or fraction. Acceptable concordance of model-predicted effects and no-effects concentrations with toxicity values derived from laboratory studies has been reported for test species for aquatic and sediment tests McGrath et al. 2005x; McGrath and Di Toro 2009; Redman et al. 2012; USEPA 2003. This section describes a few of the most commonly used predictive approaches.
A brief overview of the strengths and limitations associated with the models for prediction of TPH toxicity is provided in Table 7-7. It is important that the user be aware of the foundation and basis of the model of choice, the inherent assumptions and uncertainties, and the intended uses.
Table 7‑7. Summary of advantages and limitations for ecotoxicity modeling tools
| Description/Usage | Advantages | Limitations |
|---|---|---|
| Narcosis Target Lipid Model | ||
| Most commonly applied in sediment risk assessment | Generally accepted by agencies after discussion | There may be multiple modes of toxic action in addition to narcosis, some of which can be accounted for by applying an acute to chronic ratio (ACR). |
| PETROTOX | ||
| Primarily for surface water; can be adjusted for groundwater discharge to surface water by selection of input options; should use most current version of model and user manual | Includes acute and chronic toxicity data for 42 freshwater and saltwater test species of algae, invertebrates, and fish; uses robust species sensitivity distribution approach; can back-calculate acceptable TPH levels for any desired percentage of exposed taxa | Primarily intended to model toxicity for product-based scenarios; requires some model adjustments to apply to contaminated site evaluation; works best with high resolution fractional data; recommend discussion with agencies prior to use; not all agencies accept species sensitivity distribution approach; chronic effects are estimated more by the use of ACRs than chronic toxicity data |
| PETRORISK | ||
| Can be used to estimate multimedia ecological and human health risks for exposure to petroleum products during the product life cycle; incorporates physical/chemical model properties from PETROTOX | Similar to PETROTOX | Similar to PETROTOX |
| ECOSAR/EPI Suite | ||
| ECOSAR estimates acute and chronic toxicity based on narcosis model; uses EPI Suite to develop physical/chemical properties for fate and transport and intermedia transfer calculations | Generally accepted by agencies; includes TPH aliphatic and aromatic hydrocarbons; most useful and robust for freshwater algae, invertebrates, and fish; | Aquatic receptors only; very little information for saltwater species; requires match-up between analytical data and chemical classes included in the program |
| QSAR | ||
| Similar to ECOSAR | Accepted by agencies | Similar to ECOSAR, some QSAR models and applications have extensive learning/knowledge requirements |
7.4.3.3 Use of Indicator and Surrogate TPH Compounds
Toxicity assessment based on the concepts of indicator and surrogate chemicals is often used in risk assessment, but they are not identical and serve different purposes.
For chemical mixtures, an indicator chemical(s) is often used to account for the main toxic chemical of the mixture. That is, if the risk from the indicator chemical is acceptable, then risk from the mixture is also assumed to be acceptable. An example of this is the use of PAHs as indicator compounds for assessment of the toxicity of petroleum substances in sediments. However, the USEPA and others have demonstrated that sediment toxicity at several sites is conservatively predicted by bulk sediment PAH concentrations, and the estimation of pore water concentrations using equilibrium partitioning theory, or better still the direct measurement of freely dissolved PAH concentrations in sediment pore water, can provide a more accurate indication of toxicity to benthic organisms USEPA 2003, 2012b; Kreitinger et al. 2007; McDonough et al. 2010.
For TPH in soils, surrogate chemicals with similar chemical structures to the other chemicals in the mixture or fraction are also often used in ERAs when toxicity values for the detected chemicals are lacking. Surrogate chemicals are used to account for the toxicity of the TPH mixture or a TPH fraction. For example, USEPA developed Ecological Soil Screening Limits (Eco-SSLs) for low molecular weight (LMW) and high molecular weight (HMW) PAHs using surrogate chemical data. Naphthalene is similar to other LMW PAHs in chemical reactivity, environmental fate, and toxicological effects. Therefore, naphthalene screening levels and toxicity reference values (TRVs) were used as surrogates for all LMW PAHs and applied to the total concentration of LMW PAHs. Likewise, screening levels and TRVs developed for a HMW PAH, such as benzo(a)pyrene or 7,12-dimethylbenz(a)anthracene, were used to approximate toxicity of all HMW PAHs on equipotent basis. Although some approaches may select the most toxic chemical to represent an entire fraction, such a conservative approach may overestimate risks, especially when sufficient analytical data are available regarding the presence or absence of the selected compounds Vedagiri and Curren 2014.
7.4.3.4 Toxicity of Polar Degradation Products to Ecological Receptors
Approaches to evaluating the toxicity of polar degradation compounds are still evolving due to lack of data on their potential toxicity and persistence. For a detailed discussion about the nature of polar degradation products and challenges of evaluating their toxicity see Section 6.10.
As noted in TPH Fundamentals, polar degradation products are primarily produced by biodegradation but can also be produced by chemical degradation such as photooxidation. For ecological receptors, photooxidized products can be formed in environmental media (i.e., photomodification) or within the tissues of an organism (i.e., photosensitization), potentially resulting in phototoxicity. In this section, the focus is on new polar, photooxidized degradation products produced in environmental media that have their own inherent properties and toxicity.
Environmentally relevant studies on the ecotoxicity of polar metabolites are still developing, especially for refined product releases. Although some studies are available regarding the potential ecological toxicity of some of the polar degradation products Rogers et al. 2002; Mao et al. 2009a; Melbye et al. 2009; Neff et al. 2000; Thomas, Donkin, and Rowland 1995; Wolfe et al. 1995, only limited data are available and research is ongoing to fill some of these data gaps Lundegard and Knott 2001; Mao et al. 2009a, 2009b; Zemo, O’Reilly, et al. 2013; Zemo et al. 2017. Results from these studies suggest that overall toxicity appears to decrease with increased transport distance or with degree of weathering (time). A literature review of these studies has recently been published in a State of California guidance document CASWB-SFBR 2016b.
Two approaches are currently used to assess the toxicity of polar degradation products to ecological receptors. The first approach is to assume that polar degradation products in a TPH mixture have toxicity that is similar to or less than the toxicity of the parent hydrocarbons CASWB-SFBR 2016a; HIDOH 2017. Under this approach, sample data generated by analytical methods that do not selectively remove polar compounds (i.e., without the use of silica gel cleanup, or non-SGC TPH data) are directly compared to TPH screening levels for toxicity. This approach is typically recommended for initial site screening purposes.
The second approach is to separate polar degradation products and petroleum hydrocarbons in environmental samples and conduct toxicity testing on the extracts to assess the toxicity of site-specific mixtures. Soils and sediments contain naturally occurring polar organic compounds, particularly if the media are rich in organic material, and these compounds should be considered when evaluating the toxicity of polar degradation products in soils and sediments at petroleum release sites. A comparison of the toxicity observed at background or reference sites with similar site characteristics to that of site samples may provide information about the contribution of naturally occurring polar organic compounds to toxicity of site samples.
A third approach may be possible in the future and could reduce the need for toxicity testing of site media. This approach, as is described for human health risk assessment in Section 6, assigns toxicity factors to individual polar degradation products, which then represent the toxicity of chemical structural families of degradation products Zemo, O’Reilly, et al. 2013. The primary limitation of this approach for assessment of toxicity to ecological receptors is that data are limited to determine and assign toxicity factors that would represent the different classes of receptors or even the most common classes of ecological receptors (e.g., fish, aquatic invertebrates), but research is ongoing to fill these data gaps.
7.4.3.5 Toxicity Testing Methods
Toxicity testing methods are available to evaluate TPH toxicity in the context of releases that have already occurred, as well as to predict toxicity if a release were to occur in the future. In general, there are more established toxicity testing methods for TPH impacts to aquatic and benthic species than for terrestrial species.
7.4.3.5.1 TPH Toxicity Testing
Ecological toxicity associated with TPH can be directly evaluated using a variety of test media, species, and experimental design/conditions. A vast array of toxicity tests is available. They have been used for testing of various exposure media, such as soils, bulk sediments, surface water, pore water, WAFs, and WSFs. The nature of the source TPH may be fresh or weathered, whole products or residues, fractions or individual constituents. The test species may include terrestrial plants and seedlings, terrestrial invertebrates, such as earthworms, and aquatic and benthic species, such as algae, invertebrates, and fish. Both freshwater and saltwater test species have been used.
Given the complex mixture of TPH, interpreting toxicity tests can be challenging. Toxicity tests must be conducted such that the actual exposure concentration and composition can be measured to extrapolate the data to site-specific conditions. This is particularly important when testing the toxicity of petroleum products to aquatic organisms where the dissolved hydrocarbons in the WSF or WAF may be challenging to quantify Redman and Parkerton 2015. Although there is no one compendium of recommended test species and procedures for toxicity testing of petroleum hydrocarbons, guidelines for standardized aquatic toxicity test methods for petroleum substances have been developed and published Redman and Parkerton 2015; Aurand and Coelho 2005; Singer et al. 2000; Barron and Ka’aihue 2003.
Guidelines for toxicity testing of chemical constituents in environmental media, such as water, soil, or sediments, have been developed by numerous international and national regulatory agencies, including the OECD 2017 and USEPA 1994. In designing site-specific toxicity tests, it is useful to include reference locations (locations not impacted by the TPH release or that of other hazardous substances) to account for natural variability that might impact test results and lead to a potential false positive (i.e., conclusion that TPH release causes toxicity) in regards to test interpretation.
7.4.3.5.2 Whole Effluent Toxicity Testing
There may be concerns about TPH toxicity to aquatic biota residing in surface water if TPH-containing groundwater were to discharge into surface water bodies. Whole Effluent Toxicity (WET) testing can be performed at TPH sites to assess the spill-related aquatic toxicity of contaminated groundwater discharging to surface water CASWB-SFBR 2016a; Zemo, O’Reilly, et al. 2013; Chakrabarti 2018. WET testing measures the combined effect of a complex effluent (mixture) where it is typically infeasible to measure all individual compounds (and where single-chemical toxicity information is lacking). WET testing methods are a component of the National Pollutant Discharge Elimination System (NPDES) permit program, authorized by the Clean Water Act, that controls water pollution by regulating point source discharges into waters of the United States SETAC 2004; USEPA 1991, 2000a. Specific considerations for WET testing of TPH mixtures include testing a background groundwater sample (unimpacted by the TPH spill in an upgradient or nearby location that has similar hydrogeologic and vegetative characteristics) and monitoring the TPH concentration of the groundwater during the test protocol to evaluate whether there is significant loss through volatilization or biodegradation. Site-specific examples of WET testing can be found in the literature Chakrabarti 2018.
7.4.4 Toxicity Basis for Screening and Site-Specific ERA
There is intentional difference in the level of representativeness and conservatism associated with the toxicity criteria and values used at the screening-level and site-specific level of risk assessments.
7.4.4.1 Toxicity Basis TPH Screening Levels
Screening levels for ecological risk assessment are discussed in Section 7.2. Regulatory screening values for TPH are available for surface water, and to a lesser degree, for soil and other media. The screening levels are generally based on direct toxicity, such as protection of algae, invertebrates, and fish that may be directly exposed to TPH in surface water. Soil screening levels for the protection of terrestrial vegetation have been published in Canada CCME 2008. The sources and links provided in Tables 7-8 through 7-10 may also be checked to obtain the most current version of available toxicity data for screening levels. It is noted that the information and sources shown in these tables may change considerably over time as new data become available.
7.4.4.2 Toxicity Reference Values for Site-Specific ERA
The availability of toxicity data for TPH greatly influences the depth and breadth of ecological risk assessment that can be performed. Ecological toxicity data are available and constantly updated for various TPH fractions (see Table 7-8). The sources of information include government agencies, industry associations, academic research, and other technical and “gray” literature.
Tables 7-8 through 7-10 provide a listing of sources and relevant analytical constituents for each of the products, fractions, and constituents for which ecological toxicity information is available for both freshwater (FW) and saltwater (SW) species. This summary is based on data that were readily available online as of January 2017. Because toxicity data are constantly being updated and reinterpreted, the purpose of this table is not to provide a master compilation of toxicity reference values, but rather to identify sources where the most up-to-date information may be available for the constituent and species of interest. The information provided should be used with caution and with an understanding of the assumptions and uncertainties. It is recommended that the primary literature also be accessed and reviewed, as appropriate, by an experienced ecotoxicologist/ecological risk assessor.
Searchable databases that provide toxicity data by chemical and test species of interest are also available, including two of particular importance:
Additionally, information is also available at other sources, including:
- Oak Ridge National Laboratory maintains a comprehensive database of ecological screening values
- American Petroleum Institute’s High Production Volume (HPV) Challenge Program Testing
- NOAA’s CAFÉ: Chemical Aquatic Fate and Effects Database
The availability of ecological toxicity data varies with the type of TPH and the ecological receptor. In general, more data are available for aquatic receptors than for terrestrial receptors. Toxicity data from laboratory-based testing are more available for individual constituents (e.g., BTEX), certain chemical groups (PAHs), and whole products than for TPH fractions. Toxicity values developed for fractions (whether or not they are separated into aliphatic and aromatic fractions) may be based on the interpreted combinations of modeling and toxicity testing results where the assumed physical and chemical properties of individual fractions are applied to whole product testing to estimate fraction-specific toxicity values. Limited toxicity data are available for amphibians, birds, and mammals.
7.5 Risk Characterization
In ecological risk assessment USEPA 1997a, the risk characterization integrates the exposure (Section 7.3) and toxicity assessments (Section 7.4). For general guidance on methods and approaches to risk characterization for ecological risk assessment, see the references noted in Section 7.1.
7.5.1 Screening-Level Risk Characterization
Screening-level ecological risk assessment relies on comparison of site data against default or site-specific screening values. At the screening level, maximum or statistical estimates of TPH concentrations in the media of interest are compared to their corresponding screening levels. The lack of exceedances or the number and magnitude of exceedances of the screening levels are used to determine whether the evaluation can terminate at the screening level (i.e., no threats to ecological receptors and no further evaluation warranted) or if a site-specific risk assessment should be undertaken (i.e., there may be potential for adverse effects and additional evaluation is warranted).
7.5.2 Site-specific Level Risk Characterization
Site-specific ERA identifies and develops toxicity data and exposure parameters for identified receptors and assessment end points at the site, and estimates risk based on hazard quotients by comparing media concentrations to measured or estimated media toxicity values and/or ingested doses compared to toxicity reference values. Section 7.4 provides detailed information on toxicity criteria, methods, and approaches.
7.5.3 Multiple Lines of Evidence Approach in Site-Specific TPH ERA
Multiple Lines of Evidence (MLOE) Approach
The MLOE approach serves to increase the robustness of and level of confidence in a risk assessment. When all lines of evidence point to similar trends and findings, there is greater confidence in the overall conclusions of the ERA. In TPH ERA, the MLOE approach may include using several types of appropriate TPH data (e.g., product-level, fraction-level, and indicator compounds data) and several risk characterization methods (e.g., predicted hazard quotients, toxicity tests, and population and community-level impact metrics). When assuming that the toxicity of TPH compounds or fractions is additive, it is important to avoid “double-counting” of hazards in the assessments (see Sections 7.5 and 7.6).
Risk characterization for site-specific risk assessment introduces site-specific lines of evidence and measurement end points to address the specific nature of the TPH issues at the site. Table 7-11 presents general options and recommendations. Applying more than one line of evidence to a complex site can be helpful in understanding the potential impacts of TPH mixtures.
Table 7‑11. Key lines of evidence for site-specific TPH ecological risk characterization
| Media and Receptors | TPH Applicability | Best Practices |
|---|---|---|
| Line of Evidence: Calculation of TPH hazard quotients | ||
| Ingested dose to higher trophic level receptors in aquatic and upland environments | Relies on defining appropriate toxicity reference values and exposure parameters for the TPH exposure | Generally recognized and straightforward method. Easy to interpret. Multiple toxicity criteria from diverse sources and absence of generally applied criteria require careful identification and justification of selected values. |
| Line of Evidence: Food web uptake and biotransfer | ||
| Higher trophic level receptors (predators) indirectly exposed via prey | Limited applicability. Biotransfer of TPH difficult to evaluate. Evaluation often based on known bioaccumulative components, such as PAHs | Food web uptake is usually part of site-specific risk assessment for individual chemicals. However, not easily measured or defined for TPH and therefore not recommended except for PAHs in aquatic environments and in special circumstances. |
| Line of Evidence: Toxicity testing and bioaccumulation studies | ||
| Water and sediment: applicable and generally accepted for media-specific and receptor-specific applications
Soil: less common, but frequently applied to specific receptor classes such as rooting plants and soil invertebrates |
In surface: water toxicity testing, WAF toxicity tests.
In sediment: sediment and pore water bioassays. In soil: plant and invertebrate toxicity testing All toxicity tests subject to confounding factors and lack of specificity. |
Generally recognized and applicable approach. Toxicity testing is effective in identifying adverse effects from TPH mixtures. The selection of the appropriate toxicity test is site- and situation-specific, depending on the site of impact of TPH (sediment, surface water, soil). Interpretation of toxicity tests complicated by confounding factors and may not be highly specific to TPH, therefore well-designed test objectives and protocols are critical. |
| Line of Evidence: Population and/or community metrics | ||
| Aquatic (benthic and fish) community metrics accepted and in widespread use | Susceptible to confounding factors and lack of specificity when multiple stressors are present. | Community metrics of effect are primarily used in well-defined aquatic settings. Limited applicability to TPH risk assessment due to the lack of well-defined measures differentiating TPH-related effects from other stressors. Generally not recommended for terrestrial environments at this time. |
7.5.4 Ecological Risk Characterization Considerations for TPH
This section outlines key concepts for considering TPH in the risk characterization and summarizes them in Table 7-12 for screening-level assessments and Tables 7-13 and 7-14 for site-specific risk assessments.
Table 7‑12. Screening-level risk characterization—issues and best practices
| Screening-Level Risk Assessment |
|---|
| Issue: Exclusion criteria and need for eco risk assessment
Summary: Not all petroleum release sites require ecological evaluation. Exclusion criteria can be used in many jurisdictions to exclude evaluation of TPH impacts if certain conditions are met. (Table 5-3) Best Practices: Follow local jurisdiction regulations on ecological exclusion criteria. Consider if evaluation of indicators of TPH, such as BTEX and PAHs, may adequately address the ecological concerns. |
| Issue: Multiple modes of action
Summary: Where free product is present, the concern of non-chemical-specific adverse effects from oiling, physical contact, and indirect effects from ambient oxygen deprivation and suffocation from root blockage or deoxygenation of waters, or ingestion of bulk (acutely toxic) quantities from preening may be significant. Best Practices: Consider if direct contact with TPH free product is relevant to the site. Adverse physical effects from direct contact are generally not included in TPH screening or toxicity criteria. Site-specific evaluation may be more useful for physical effects. In the absence of free product, the use of standard or modeled screening levels (for screening level) and toxicity reference values (for site-specific risk) is likely to be adequate to address the ecological risk of TPH. If free product is present, site-specific evaluation will likely be more appropriate in addition to standard dose-based evaluation, especially in aquatic risk assessment where floating, emulsified, entrained, or undissolved TPH may be present and resulting in direct toxicity effects. |
| Issue: Defining ecological screening-level criteria
Summary: There are relatively few ecological screening levels for TPH, as discussed in Section 7.2. These screening levels are frequently nonspecific as to the specific mixture or product under evaluation or are based on non-risk-based criteria. Best Practices: The options for initial screening include screening against generic TPH criteria, recognizing that the screening may say little about ecological risk, and proceed to additional site-specific tiers to further evaluate the issue if found to be exceeded. Screening analytical data against individual components of the TPH mixture, such as PAHs for heavier TPH and BTEX for lighter TPH, may be an option or requirement in certain jurisdictions. This practice does not represent best practice, as it underestimates risk from other components of the mixture but may be appropriate in limited circumstances. |
| Issue: Non-risk-based criteria
Summary: Screening levels for TPH may not be based on risk-based considerations. Such screening levels do not provide any information on whether there is an ecological risk, and it should not be assumed that there is ecological risk solely on such considerations. Best Practices: The source and relevance of screening levels should be reviewed. The use of TPH screening levels based on regulatory cleanup limits or on ad hoc values based on human health or analytical method and narrative criteria, such as the absence of visible sheens, may be appropriate depending on jurisdiction, but risk characterization should consider that it could under- or overestimate ecological risk. |
Table 7‑13. Site-specific risk characterization—issues and best practices for toxicity criteria
| Site-Specific Risk Assessment |
|---|
| Issue: Aquatic toxicity criteria and approaches
Summary: There is limited toxicity data for complete hydrocarbon mixtures, and these are focused on site-specific evaluation of marine exposures to WAFs. Models such as PETROTOX are limited to data for individual chemicals and fine-grained hydrocarbon “blocks” (see Overview of Toxicity Prediction by Mechanistic Models). The use of exposure models in aquatic systems can be a powerful tool. Best Practices: Use of toxicity screening levels based on site-specific TPH conditions is growing in acceptability, especially at petroleum release sites, and can be a powerful tool where jurisdictions accept the approach. Specialized analytical data may be required for these approaches. See Section 7.4.3 for more information on the use of such models. |
| Issue: Use of tissue criteria
Summary: The evaluation of biological tissue concentrations (e.g., bird eggs, muscle tissue, or liver concentrations) is sometimes used in site-specific ecological risk assessment. Although this has wide applicability for many chemicals, its applicability to TPH is questionable. Bioavailability and biochemistry of the components of a TPH mixture vary widely, and no single “TPH” measure can be defined or considered to be applicable across the food chain. It is conceptually difficult to posit a biochemical mode of action for TPH. TPH as an analytical end point is difficult to analyze and interpret in biological tissue due to interference from the organism’s lipids and biochemical pathways and large variations in metabolism of TPH components in biological tissue. Best Practices: It is recommended that TPH measurements in tissue not be considered in ecological risk assessment. Evaluation of specific components, such as PAHs, may be appropriate, especially where the concern is bioaccumulation and impacts to higher trophic-level organisms, but does not necessarily represent TPH as a whole. |
Table 7‑14. Site-specific risk characterization—issues and best practices for TPH TRVs
| Issue: Defining Appropriate TRVs |
|---|
| Summary: The partitioning models for TPH in aquatic systems are inapplicable or insufficient in ecological risk assessment for terrestrial receptors (exposures to soil and the terrestrial food chain) and aquatic food webs (e.g., aquatic mammals exposed to the aquatic food chain). Evaluation of ecological risk requires the definition of applicable TRVs to allow the ingested dose line of evidence to be developed. Relatively few toxicity studies, based on a low number of individual chemicals and tested on a few standard test organisms (predominantly rats and mice) under laboratory conditions, serve as the base for TPH. Therefore, TPH TRVs for the entire mixture may rely on just a few toxicity study results, implying that the entire mixture or fraction has equivalent and additive toxicity as the test compound used as surrogate.
Best Practices: Best practice for using TPH TRVs in site-specific risk assessment may be ranked as follows:
|
7.5.5 Uncertainty Analysis
Uncertainties are integral to all risk assessment and particularly so for ecological risk assessment, considering the variability of real-world ecological systems and the unknowns inherent in evaluating a large class of chemicals as is the case with TPH. In the uncertainty section, the risk characterization results are evaluated in terms of confidence in the outcome based on the uncertainties, confidence levels, and applicability of the conceptual site model, exposure assumptions, toxicity assumptions, and underlying data. The evaluation of uncertaintiesan be narrative and may include qualitative or quantitative sensitivity analysis based on alternate assumptions to bound the estimates.